*Base substitutions, insertions or deletions, copy number alterations and gene rearrangements.
†309 genes with complete coding exonic coverage, 15 genes with select intronic or non-coding regions only.
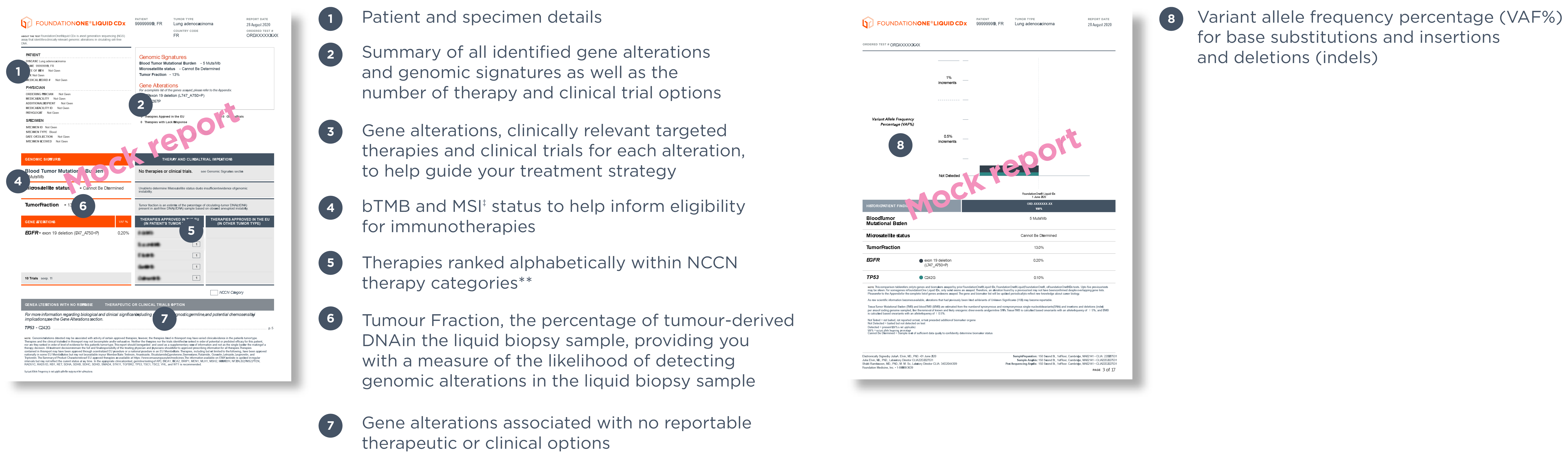
‡Foundation0ne® Liquid CDx reports MSI-H status
§Validation based on overall >7,500 samples covering >30,000 unique variants across >300 genes and 37 cancer indications.1,2 To learn more about the clinical and analytical validation of FoundationOne® Liquid CDx, click here.
¥75 genes are baited with enhanced sensitivity for all variant types (selected based on increased actionability with current or future targeted therapies; for more information of these 75 genes, please refer to our full gene list): other genomic regions are baited with high sensitivity.
¶Clinical validation based on evidence gathered using an earlier version of Foundation Medicine's current liquid biopsy service, FoundationOne® Liquid CDx. For concordance results between these two tests, please see our full intended use at www.foundationmedicine.com/F1LCDx.
**For additional information on the NCCN categories please refer to the NCCN Compendium® (www.nccn.org).
bTMB, blood Tumour Mutational Burden; cfDNA, circulating cell-free DNA; CGP, comprehensive genomic profiling; ctDNA, cell-free tumour DNA; d, days; FDA, US Food and Drug Administration; MSI, Microsatellite Instability; NCCN, National Comprehensive Cancer Network; NGS, next generation sequencing; NSCLC, non-small cell lung cancer.